
The VivAer Stylus
Helping Patients Find Lasting Relief from Nasal Airway Obstruction (NAO)
Millions of people suffering from nasal airway obstruction struggle with their quality of life.1 Many are actively seeking a proven, long-term solution to breathe easier.
A Trusted Solution for Your NAO Patients
VivAer offers a non-invasive, incision-free procedure for nasal airway obstruction, providing an effective in-office treatment option. The compact and portable Aerin® Console and VivAer Stylus easily integrate into any clinic or hospital setting, making it a convenient and efficient choice for your practice.
Treat the Multiple Sources of NAO
Small Adjustments May Lead to Significant Results
The nasal valve, responsible for two-thirds of nasal airway resistance, is the narrowest part of the nasal passage. According to Poiseuille’s Law, airflow increases exponentially with even small changes to the nasal airway, leading to noticeable improvements in breathing. VivAer utilizes temperature-controlled radiofrequency technology tailored for the nasal airway, enabling you to treat the lateral nasal wall, full length of the inferior turbinate, and septal swell body in a single, efficient session.
Proven Outcomes
VivAer is backed by extensive clinical research, demonstrating long-term relief for nasal airway obstruction (NAO). In a multicenter study of patients treated with VivAer, participants consistently reported improved nasal breathing, even during exercise and physical exertion.
Increased Access for VivAer Patients
VivAer is covered by qualifying insurance plans when deemed medically necessary by a healthcare provider or ENT. The Aerin® Reimbursement Center (ARC) is a HIPAA compliant, complimentary, third-party reimbursement support program made available by Aerin® Medical to assist physicians and their patients with VivAer insurance verification.
Lateral Nasal Wall
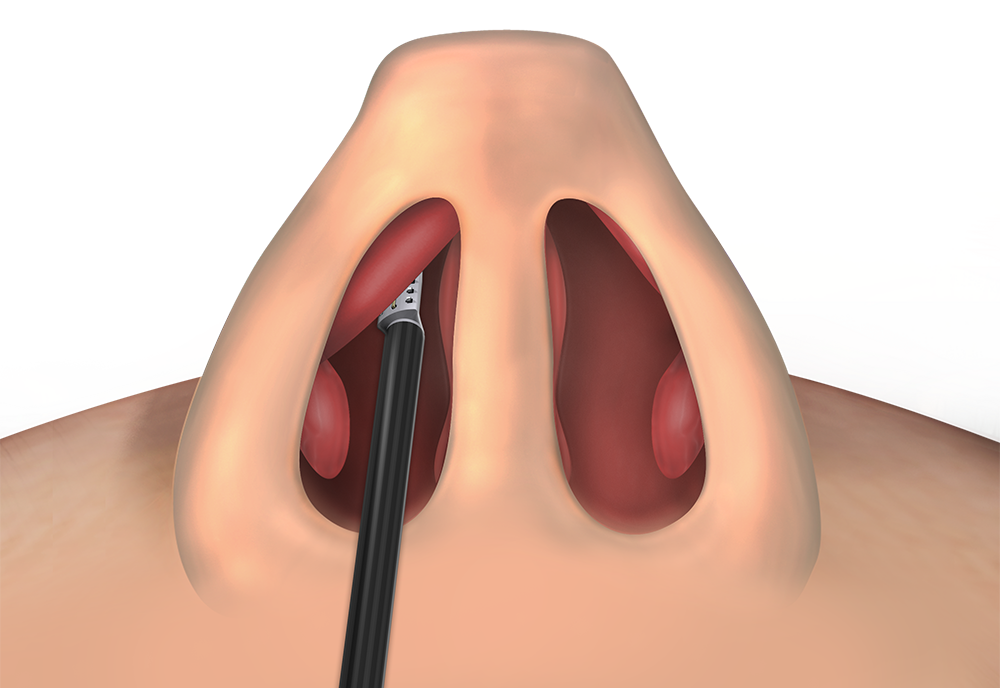
CPT® 30469
Repair of nasal valve collapse with low energy, temperature-controlled (i.e., radiofrequency) subcutaneous/submucosal remodeling
Septal Swell Bodies
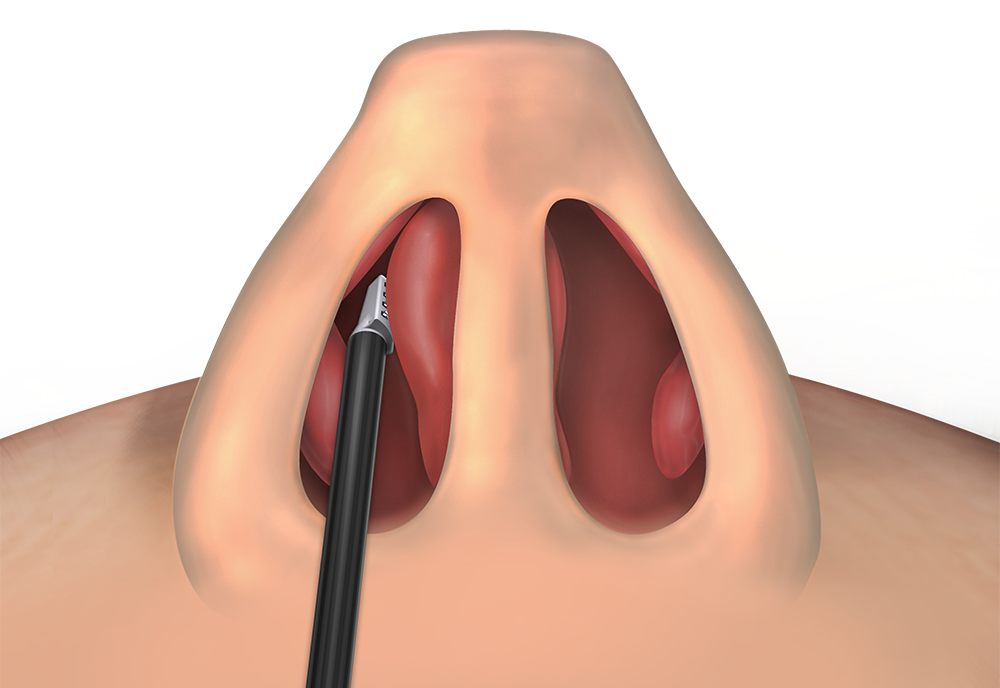
CPT 30117
Excision or destruction (e.g., laser),
intranasal lesion; internal approach
Inferior Turbinate
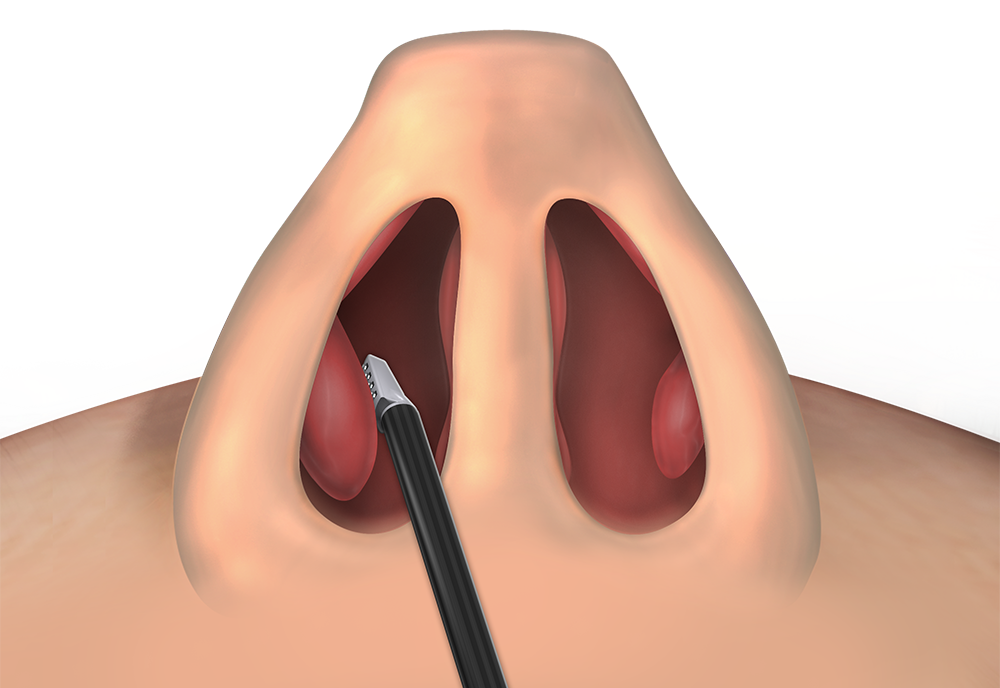
CPT 30802 or CPT 30801
Ablation, soft tissue of inferior turbinates, unilateral or bilateral, any method (volume reduction). Submucosal (CPT 30802) or superficial (CPT 30801)*
Disclaimer
The VivAer® Stylus is indicated for use in otorhinolaryngology (ENT) surgery for the coagulation of soft tissue in the nasal airway, to treat nasal airway obstruction by shrinking submucosal tissue, including cartilage in the internal nasal valve area. Specific indications, contraindications, warnings, precautions, and safety information exist for these products.
Aerin Medical relies on the physician to determine, assess and communicate to each patient all foreseeable risks of the procedure. A physician must always refer to the package insert, product label and/or instructions for use before using any of Aerin Medical’s products. Rx only.
Current Procedural Terminology (CPT) Copyright 2025 American Medical Association (AMA). All rights reserved. CPT® is a registered trademark of the American Medical Association.
References:
- Stewart MG, Ferguson B, Fromer L. Epidemiology and burden of nasal congestion. Int J Gen Med. 2010;3:37-45. https://doi.org/10.2147/IJGM.S8077
- 4-Year NV Pivotal Study Clinical Summary Report is on file with the company. MKT1469.A
- Jacobowitz, O, Ehmer, D, Lanier, B, Scurry, W, Davis, B. Longterm outcomes following repair of nasal valve collapse with temperature-controlled radiofrequency treatment for patients with nasal obstruction. Int Forum Allergy Rhinol. 2022; 1- 5. https://doi.org/10.1002/alr.23019
- Provider to determine appropriate coding for destruction of turbinate either superficial or intramural (i.e., submucosal).
WEB1322-04.H





